Chlorine has become inseparable from modern water treatment. It is effective, affordable, and widely understood. For disinfection, it works. For distribution safety, it works. For regulatory compliance, it works.
But chlorine is rarely meant to remain in water beyond a certain point.
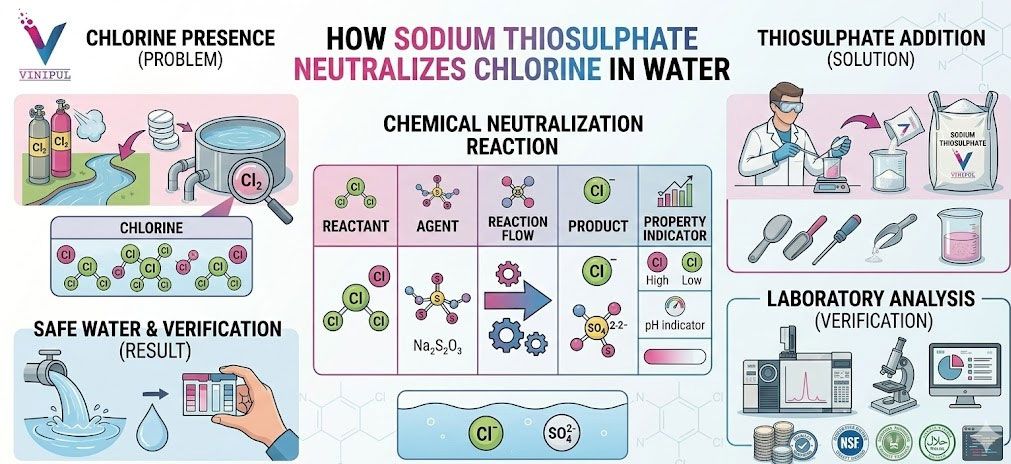
In real plants—STPs, ETPs, tertiary treatment units, reuse systems—residual chlorine becomes a liability. It damages downstream biological processes, harms aquatic life, corrodes equipment, and fails discharge norms if not controlled.
This is where sodium thiosulphate enters the picture.
Not as a disinfectant. Not as a polishing chemical. But as a chemical stop-button—a way to decisively neutralize chlorine once it has done its job.
Most operators know that sodium thiosulphate “removes chlorine.” Fewer understand how it does so, why it is preferred over other dechlorinating agents, and what really happens inside the water when it is dosed.
This article explains that process from an operational, plant-level perspective—without lab jargon and without oversimplification—so decision-makers understand not just that sodium thiosulphate works, but why it is trusted across water treatment systems worldwide.
What Sodium Thiosulphate Does — Explained Practically
In water treatment, sodium thiosulphate functions as a reducing agent.
That single phrase matters.
Chlorine is an oxidizing agent. Its disinfecting power comes from its ability to oxidize microorganisms and organic matter. Sodium thiosulphate neutralizes chlorine by chemically reducing it—converting active chlorine species into harmless chloride ions.
From an operational point of view, sodium thiosulphate:
- Reacts quickly
• Reacts predictably
• Leaves no toxic by-products
• Does not introduce aggressive chemistry
This makes it particularly suitable for controlled, final-stage water treatment where precision matters more than brute force.
Unlike physical removal methods, sodium thiosulphate destroys chlorine chemically rather than separating it.
Understanding Chlorine in Water — Why Neutralization Is Required
Chlorine exists in water in several active forms:
- Free chlorine (Cl₂, HOCl, OCl⁻)
• Combined chlorine (chloramines)
These forms are intentionally maintained during disinfection. But beyond that stage, they create problems.
Problems Caused by Residual Chlorine
In real-world plants, residual chlorine can:
- Kill beneficial bacteria in biological treatment stages
• Damage membranes in RO and UF systems
• Fail discharge limits for surface water release
• Harm aquatic ecosystems
• Accelerate corrosion in pipelines
Because of this, many regulations require zero or near-zero chlorine residual before discharge or reuse.
Neutralization is not optional. It is a compliance step.
The Chemical Mechanism — How Sodium Thiosulphate Neutralizes Chlorine
The Core Reaction (Conceptual)
When sodium thiosulphate is added to chlorinated water, it reacts with chlorine through a redox reaction.
In simple terms:
- Chlorine is reduced to chloride (Cl⁻)
• Thiosulphate is oxidized to sulfate
The chlorine loses its oxidative power. Once reduced to chloride, it becomes chemically inert in the context of disinfection and toxicity.
What matters operationally is not the equation itself, but the outcome:
- No free chlorine remains
• No disinfecting activity remains
• No toxic residual is introduced
This is why sodium thiosulphate is widely accepted by regulators.
Why This Reaction Is Preferred
The reaction between sodium thiosulphate and chlorine is:
- Fast (seconds, not minutes)
• Complete (no partial neutralization)
• Stoichiometrically predictable
This predictability allows engineers to calculate dosage precisely, even in fluctuating flow conditions.
Why Sodium Thiosulphate Is Preferred Over Other Dechlorinating Agents
Several chemicals can neutralize chlorine. Sodium thiosulphate is not the only option—but it is often the most practical.
Compared to Sulphur Dioxide or Sulphites
Sulphur-based gases and sulphites can neutralize chlorine, but they introduce issues:
- Handling risk
• Toxic gas exposure
• Over-reduction leading to oxygen depletion
• Stricter safety protocols
Sodium thiosulphate avoids these risks while delivering equivalent neutralization efficiency.
Compared to Activated Carbon
Activated carbon removes chlorine by adsorption, not chemical reaction.
Limitations include:
• Media exhaustion
• Channeling
• Higher capital cost
• Maintenance complexity
Sodium thiosulphate offers instant chemical certainty without mechanical dependency.
Compared to Sodium Metabisulphite (SMBS)
SMBS is effective but more aggressive.
Challenges include:
• Sulphite residual concerns
• Oxygen scavenging
• Higher sensitivity to overdosing
Sodium thiosulphate is more forgiving, especially in municipal and mixed-use systems.
Where Sodium Thiosulphate Is Used in Water Treatment Systems
Municipal Water Treatment Plants
Used to:
• Neutralize residual chlorine before discharge
• Protect aquatic environments
• Comply with environmental norms
Municipal operators value sodium thiosulphate because it is easy to dose and safe to handle.
Sewage Treatment Plants (STP)
In STPs, chlorine is often used for:
• Final disinfection
• Odor control
Sodium thiosulphate is applied downstream to:
• Protect receiving water bodies
• Prevent biological toxicity
Effluent Treatment Plants (ETP)
Industrial effluents often carry variable chlorine loads.
Sodium thiosulphate is preferred because:
• Dosage can be adjusted quickly
• Reaction remains stable across pH ranges
• No secondary contamination is introduced
RO / Membrane Protection
Chlorine damages membranes irreversibly.
Sodium thiosulphate is used upstream of:
• RO systems
• UF membranes
to ensure chlorine does not reach sensitive equipment.
Dosage Logic — How Operators Determine Quantity
Sodium thiosulphate dosing is not guesswork.
Operators consider:
• Chlorine concentration (ppm)
• Flow rate
• Contact time
The reaction ratio is well established, allowing precise calculation.
Key operational principle:
Dose only what is required to neutralize chlorine—nothing more.
Overdosing does not improve safety. It only increases cost and sulfate load.
Operational Advantages in Real Plants
From years of plant operation experience, sodium thiosulphate offers:
- Stable storage
• Easy solubility
• Low corrosion potential
• Minimal safety hazards
It does not release fumes, does not require pressurized systems, and does not demand specialized handling beyond basic chemical safety.
This matters in plants where staffing and training levels vary.
Quality & Grade Considerations
Not all sodium thiosulphate performs the same.
Bulk buyers look for:
• Consistent purity
• Controlled moisture content
• Low insoluble matter
Impurities can:
• Interfere with dosing accuracy
• Create downstream scaling
• Reduce reaction efficiency
In large plants, consistency matters more than headline purity numbers.
Bulk Supply & Commercial Reality
For large STP and ETP projects, sodium thiosulphate is procured in bulk.
Commercial considerations include:
• Monthly production capacity
• Packaging suitability
• Logistics reliability
Plants running continuously cannot afford inconsistent supply. This is why operators prefer suppliers who treat sodium thiosulphate as a process chemical, not a trading commodity.
Common Operational Mistakes
Experienced engineers see the same mistakes repeatedly:
- Treating dechlorination as optional
• Under-dosing to “save chemical”
• Switching dechlorinating agents without recalculation
• Ignoring seasonal chlorine variation
Sodium thiosulphate works best when applied as part of a controlled system, not as an emergency fix.
FAQ
How does sodium thiosulphate neutralize chlorine?
By chemically reducing chlorine into harmless chloride ions.
Is sodium thiosulphate safe for water treatment?
Yes, when used correctly it leaves no toxic residues.
Does sodium thiosulphate remove chloramines?
Yes, though dosage requirements differ from free chlorine.
Can sodium thiosulphate damage equipment?
No. It is far less corrosive than many alternatives.
Is overdosing a problem?
It increases cost and sulfate load but does not reintroduce toxicity.
Why is it preferred in STPs and ETPs?
Because it is predictable, safe, and regulator-friendly.
Final Perspective
Sodium thiosulphate is not used in water treatment because it is fashionable or inexpensive. It is used because it ends chlorine activity cleanly and decisively.
Chlorine is excellent at disinfection. Sodium thiosulphate is excellent at knowing when to stop it.
That balance—use chlorine where it protects, neutralize it where it harms—is what modern water treatment is built on.
Plants that understand this treat dechlorination as a core process step, not a checkbox. And in that role, sodium thiosulphate has earned its place as one of the most trusted chemicals in water treatment operations worldwide.