Sodium phosphates sit quietly inside hundreds of industrial processes. They are rarely the hero ingredient, rarely the most expensive line item, and rarely discussed unless something goes wrong.
Yet when the wrong sodium phosphate is selected, the consequences show up fast—failed audits, unstable formulations, inconsistent product quality, rejected export consignments, or unexpected downtime.
Most buyers don’t struggle to source sodium phosphates. They struggle to choose the right one.
Mono, di, tri. Food grade, technical grade. Buffered, alkaline, reactive. On paper, many sodium phosphates look interchangeable. In real operations, they are not.
This confusion usually appears when:
- A plant expands into a new product category
- An export customer introduces stricter compliance checks
- A formulation behaves differently after a supplier change
- A QA audit questions grade justification
At that point, the decision stops being commercial and becomes technical.
This guide explains how experienced buyers and process owners choose the right sodium phosphate for their industry—not by habit or price alone, but by understanding function, risk, and long-term operational impact.
2️⃣ What Sodium Phosphates Do — Explained Practically
Sodium phosphates are used because they control conditions inside a process.
Depending on the type, they can:
- Buffer pH
- Bind water
- Disperse solids
- Stabilize emulsions
- Improve cleaning efficiency
- Prevent scale formation
What changes from one sodium phosphate to another is how aggressively and where they perform these roles.
This is why choosing the right sodium phosphate is less about chemistry theory and more about process behavior.
3️⃣ The Three Core Sodium Phosphates — Practical Differences
Mono Sodium Phosphate (MSP)
Mono sodium phosphate is mild and acidic.
From an operational standpoint:
- It adjusts pH gently
- It reacts predictably
- It rarely destabilizes formulations
It is chosen when:
- Fine pH control is needed
- Sensory impact must be minimal
- The process is sensitive to alkalinity
MSP is often the safest option when uncertainty exists.
Di Sodium Phosphate (DSP)
Di sodium phosphate is balanced and versatile.
In real plants, DSP:
- Buffers without aggression
- Integrates well across formulations
- Offers wide regulatory acceptance
This makes DSP the default choice in many industries, especially where one material must serve multiple roles.
Tri Sodium Phosphate (TSP)
Tri sodium phosphate is strongly alkaline and reactive.
Operationally:
- It delivers strong cleaning and dispersing action
- It alters protein structures quickly
- It has a narrower margin for error
TSP is chosen deliberately—not casually—and only where its strength is required.
4️⃣ Why “One Sodium Phosphate for All Uses” Fails at Scale
Smaller operations often try to standardize on one sodium phosphate to simplify procurement. At scale, this strategy usually backfires.
Reasons include:
- Different products require different pH behavior
- Regulatory limits vary by application
- Over-strength phosphates introduce instability
- Under-strength phosphates fail functionally
Experienced buyers accept that phosphate selection is application-specific, not brand-specific.
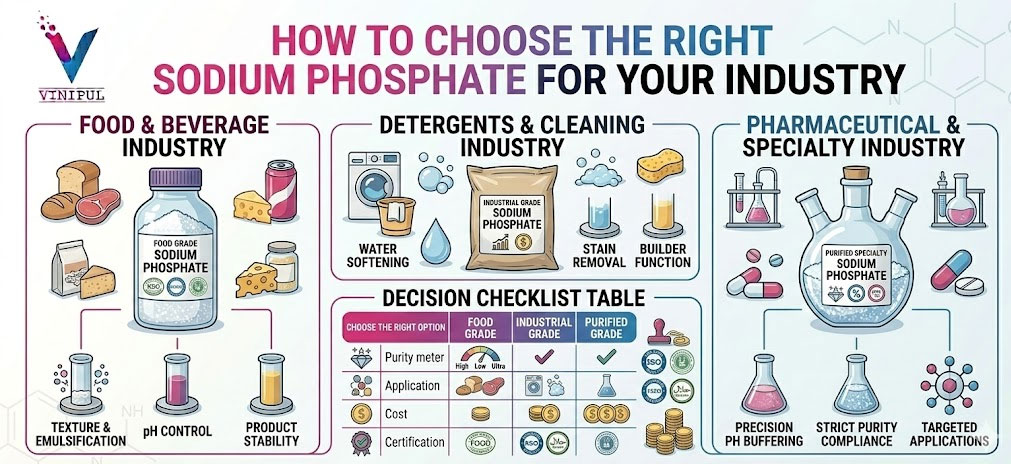
5️⃣ Industry-Wise Sodium Phosphate Selection Logic
Food & Beverage Processing
In food systems, sodium phosphates influence:
- Texture
- Water retention
- Emulsion stability
- Taste perception
Preferred choices
- MSP for beverages and bakery
- DSP for dairy, meat, and processed foods
Why:
- Gentle behavior
- Easier compliance justification
- Lower sensory risk
TSP is generally avoided in food due to its aggressive alkalinity.
Detergent & Cleaning Chemical Manufacturing
Detergent formulations demand:
- Strong alkalinity
- Soil dispersion
- Hardness control
Preferred choices
- TSP for heavy-duty detergents
- DSP in balanced formulations
Here, strength is an advantage—but only when controlled.
Water Treatment & Industrial Utilities
In water systems, sodium phosphates are used for:
- Scale control
- Corrosion inhibition
- pH adjustment
Preferred choices
- DSP for general buffering
- MSP where precise pH control is required
Over-alkalinity can worsen scaling, so TSP is used selectively.
Pharmaceutical & Nutraceutical Manufacturing
Pharma applications prioritize:
- Purity
- Predictability
- Documentation discipline
Preferred choices
- MSP and DSP (pharma grade)
TSP is rarely used due to its reactive nature and validation complexity.
Textile, Paper & Specialty Chemicals
These industries value:
- Dispersing ability
- Process robustness
Preferred choices
- DSP for versatility
- TSP for specific cleaning or dispersion stages
Selection depends heavily on downstream process sensitivity.
6️⃣ Food Grade vs Technical Grade — Why the Distinction Matters
Grade selection is often where buyers make costly mistakes.
Food Grade Sodium Phosphates
- Controlled impurity limits
- Higher documentation requirements
- Suitable for direct or indirect food contact
Technical / Industrial Grade
- Broader impurity tolerance
- Not acceptable for food or pharma
- Often cheaper
Using the wrong grade is not a minor issue—it is a compliance failure.
7️⃣ Comparison Table — Practical Buyer View
| Parameter | MSP | DSP | TSP |
|---|---|---|---|
| pH impact | Mild | Moderate | Strong |
| Ease of control | High | High | Lower |
| Food suitability | Excellent | Excellent | Limited |
| Cleaning strength | Low | Moderate | High |
| Process risk | Low | Low | Higher |
| Typical buyer profile | Food, pharma | Multi-industry | Detergent, heavy industry |
This table explains why DSP often becomes the “default” choice—it balances capability and safety.
8️⃣ How Experienced Buyers Actually Decide
They don’t start with:
“Which sodium phosphate is cheapest?”
They start with:
- What function must this phosphate perform?
- What happens if it over-performs?
- What happens if it under-performs?
- How sensitive is the process?
Only after that do they evaluate price and supply.
9️⃣ Export & Multi-Market Considerations
Buyers supplying multiple countries must consider:
- Varying food additive rules
- Labeling requirements
- Sodium intake regulations
DSP and MSP are preferred for exports because:
- They face fewer regulatory objections
- They simplify documentation
- They reduce reformulation risk
TSP often complicates export approvals.
🔟 Quality, Consistency & Supplier Discipline
Choosing the right sodium phosphate is meaningless if:
- Batch consistency varies
- Insoluble matter fluctuates
- Moisture levels are uncontrolled
Bulk buyers prioritize suppliers who:
- Maintain consistent specs
- Provide traceable documentation
- Support long-term contracts
At scale, supplier discipline matters as much as product selection.
1️⃣1️⃣ Bulk Supply & Commercial Reality
Large operations think in:
- Total cost of ownership
- Downtime risk
- Audit exposure
Using a slightly more expensive but stable sodium phosphate often saves money long-term by:
- Reducing rework
- Avoiding compliance failures
- Preventing formulation drift
1️⃣2️⃣ Common Buyer Mistakes
Repeated mistakes include:
- Treating sodium phosphates as interchangeable
- Using TSP where DSP is sufficient
- Ignoring grade requirements
- Switching suppliers without revalidation
These mistakes are usually discovered during audits—not trials.
1️⃣3️⃣ Human-Style FAQ (Voice Search Focus)
How do I choose the right sodium phosphate?
Start with function, not price. Match phosphate strength to process sensitivity.
Which sodium phosphate is safest?
MSP and DSP are safest across most industries.
Is tri sodium phosphate bad?
No, but it must be used deliberately.
Can one sodium phosphate serve all uses?
Rarely at scale.
Is food grade always required?
Yes for food, pharma, and export-sensitive uses.
Why do many buyers prefer DSP?
Because it balances performance and control.
🎯 Final Perspective
Choosing the right sodium phosphate is not about chemistry preference—it is about risk management.
At industrial scale, the best phosphate is the one that:
- Does its job quietly
- Stays within regulatory comfort zones
- Behaves consistently across batches
- Does not force downstream corrections
For many industries, that means mono or di sodium phosphate. For others, tri sodium phosphate earns its place—but only when its strength is truly required.
The mistake is not choosing the wrong phosphate once. The mistake is choosing without understanding why.
Buyers who approach sodium phosphate selection with process logic rather than habit rarely need to revisit the decision. Those who don’t usually learn the difference the hard way.